1. Why this matters for MRCGP
- Type 2 diabetes is an AKT favourite because the answer often changes with symptoms, HbA1c validity, comorbidity, kidney function, hypoglycaemia risk and driving.
- In SCA, it commonly appears as a new diagnosis, poor control, medication escalation, sick-day advice, foot symptoms, eye symptoms or a worried patient living with obesity.
- The main GP risks are missing diabetic ketoacidosis or hyperosmolar hyperglycaemic state, diagnosing from an invalid HbA1c, unsafe prescribing, and failing to organise complication screening.
- Practical use matters because diagnosis confirmation, medicine sequencing, sick-day rules, glucose monitoring, foot risk referral and DVLA advice all change safe GP action.
2. GP Bottom Line
- Recognise persistent hyperglycaemia, but do not diagnose an asymptomatic adult from one abnormal result.
- Urgency changes if the patient is acutely unwell, ketotic, dehydrated, confused, vomiting, has severe hyperglycaemia, sudden visual loss, or an active diabetic foot problem.
- Usual GP action is to confirm the diagnosis, agree an individualised care plan, offer structured education, start supported lifestyle and medicine management, and arrange complication screening.
- Exam trap: routine self-monitoring of capillary blood glucose is not advised for everyone with type 2 diabetes.
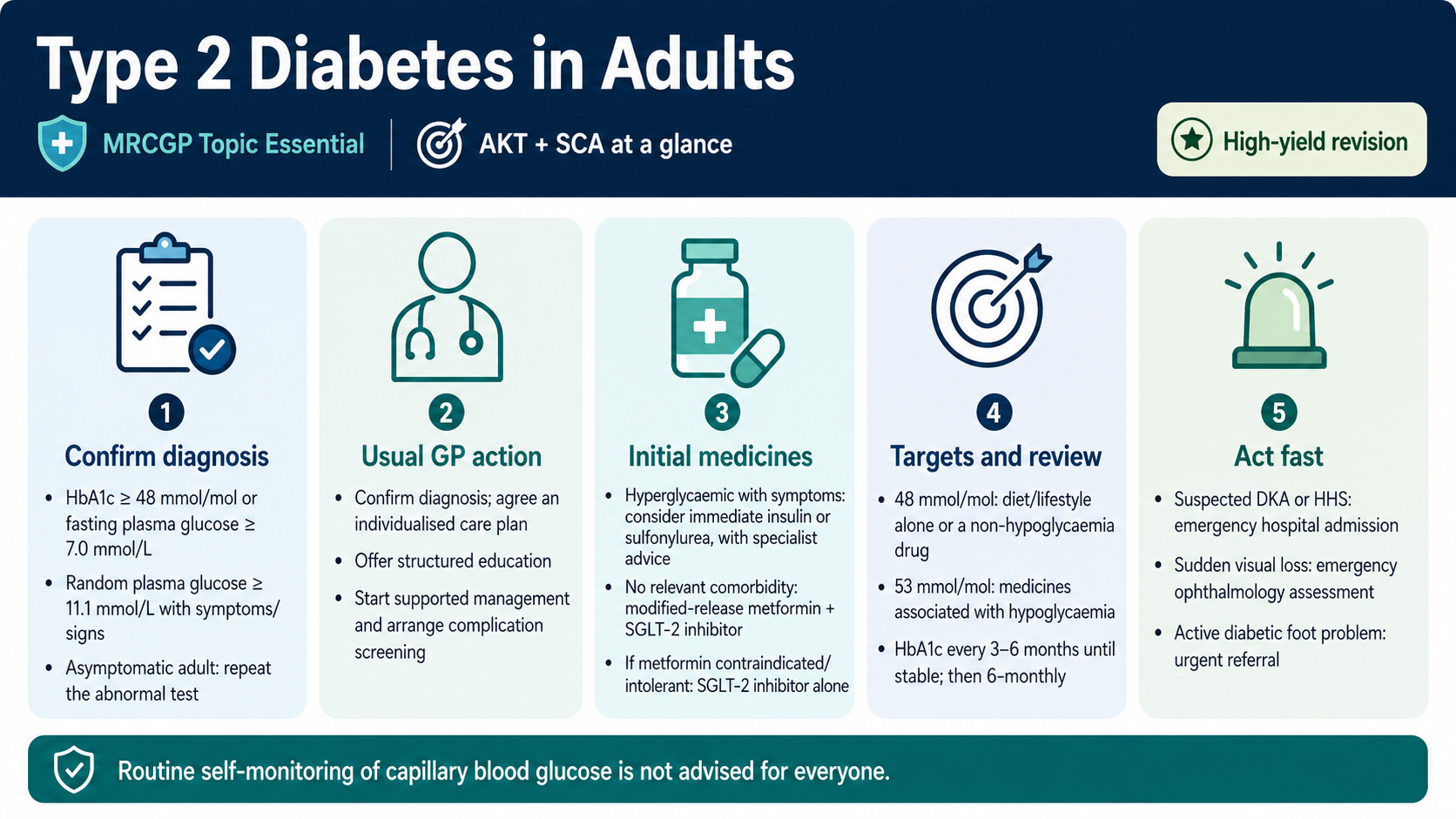
3. 60 Second Exam Snapshot
- Adult diagnostic thresholds include HbA1c 48 mmol/mol or more, fasting plasma glucose 7.0 mmol/L or more, or random plasma glucose 11.1 mmol/L or more with symptoms or signs.
- If symptomatic, one abnormal HbA1c or fasting plasma glucose can be used, though repeat testing is sensible; if asymptomatic, repeat testing is needed.
- HbA1c targets are usually 48 mmol/mol for diet/lifestyle alone or a drug not linked with hypoglycaemia, and 53 mmol/mol for medicines associated with hypoglycaemia.
- Measure HbA1c every 3–6 months until stable, then every 6 months.
- Initial adult medicine choice depends on comorbidity: modified-release metformin plus a sodium–glucose cotransporter-2 (SGLT-2) inhibitor is central in the 2026 adult pathway.
- Suspected diabetic ketoacidosis (DKA) or hyperosmolar hyperglycaemic state (HHS) needs emergency hospital admission.
- At annual review, check urine albumin:creatinine ratio, HbA1c, blood pressure, foot risk, creatinine, cholesterol, body mass index, smoking status and eye-screening status and attendance. Refer to the local eye screening service at diagnosis. Screening is every 2 years for people at low risk of sight loss after two successive screening tests with no diabetic retinopathy, and at least annually for others.
4. Recognition and Diagnosis
- Suspect type 2 diabetes in an adult with persistent hyperglycaemia, especially with polydipsia, polyuria, blurred vision, unexplained weight loss, recurrent infections, tiredness or acanthosis nigricans. Symptoms may be mild or absent.
- Risk factors include obesity and inactivity, family history, Asian, African and Afro-Caribbean ethnicity, history of gestational diabetes, polycystic ovary syndrome, metabolic syndrome, long-term corticosteroids and some other drug treatments.
- Do not over-rely on HbA1c. It should not be used to diagnose diabetes in pregnancy or within 2 months postpartum, symptoms for less than 2 months, acute illness in someone at high diabetes risk, people taking medication that may cause hyperglycaemia, acute pancreatic damage, end-stage renal disease, or people under 18. Interpret cautiously with abnormal haemoglobin, severe anaemia, altered red cell lifespan or recent transfusion.
- Consider alternatives or additional pathology if the pattern is atypical. Type 2 diabetes is more likely when there are no additional features of type 1 diabetes and no features suggesting monogenic or secondary diabetes.
- In people aged 60 or over with weight loss and any of new-onset diabetes, diarrhoea, back pain, abdominal pain, nausea, vomiting or constipation, consider an urgent direct-access CT scan, or urgent direct-access ultrasound if CT is not available. In people aged 40 or over with jaundice, use a suspected cancer pathway referral for pancreatic cancer.
5. AKT Essentials: What Changes the Answer
Diagnosis / recognition
- Symptomatic adult: one abnormal HbA1c or fasting plasma glucose may be enough, but repeat testing is sensible.
- Asymptomatic adult: repeat the abnormal test, preferably the same test.
- Severe hyperglycaemia during acute infection, trauma, circulatory stress or other stress may be transitory and is not diagnostic.
Investigation / interpretation
- If HbA1c is difficult to interpret, use fasting plasma glucose.
- If HbA1c monitoring is invalid, blood glucose trends can be assessed using plasma glucose profiles, total glycated haemoglobin estimation if abnormal haemoglobins, or fructosamine estimation.
- Unexplained discrepancy between HbA1c and glucose readings should prompt specialist diabetes or clinical biochemistry advice.
Management / next best step
- Offer structured education at or around diagnosis, with annual reinforcement and review.
- Adults should have individualised care based on preferences, comorbidity, frailty, polypharmacy and expected benefit.
- Introduce multiple medicines stepwise, checking tolerability and effectiveness.
Initial adult medicine pathway
If an adult with type 2 diabetes is hyperglycaemic with symptoms, consider immediate insulin therapy or a sulfonylurea, depending on specialist advice, and review treatment once blood glucose control has been achieved.
| Adult situation | Initial medicine choice supported by supplied adult guidance |
| No relevant comorbidity | Modified-release metformin plus SGLT-2 inhibitor; if metformin not tolerated or contraindicated, SGLT-2 inhibitor alone |
| Heart failure | Modified-release metformin plus SGLT-2 inhibitor; if no metformin, SGLT-2 inhibitor alone |
| Atherosclerotic cardiovascular disease | Modified-release metformin plus SGLT-2 inhibitor plus subcutaneous semaglutide (Ozempic) up to 1 mg once weekly |
| Chronic kidney disease, estimated glomerular filtration rate above 30 | Modified-release metformin plus SGLT-2 inhibitor |
| Chronic kidney disease, estimated glomerular filtration rate 20–30 | Dapagliflozin or empagliflozin plus a dipeptidyl peptidase-4 inhibitor |
| Chronic kidney disease, estimated glomerular filtration rate below 20 | Consider a dipeptidyl peptidase-4 inhibitor |
| Frailty | Modified-release metformin; use SGLT-2 inhibitor only if frailty does not create unacceptable volume depletion or hypotension risk |
| Early-onset diabetes, diagnosed before 40 | Modified-release metformin plus SGLT-2 inhibitor; consider glucagon-like peptide-1 receptor agonist or tirzepatide |
| Living with obesity | Modified-release metformin plus SGLT-2 inhibitor. Consider adding a GLP-1 receptor agonist or tirzepatide only if they have been taking initial therapy for at least 3 months, further medicines are needed to reach their individualised glycaemic target, and they are not already taking a GLP-1 receptor agonist or tirzepatide |
Follow-up / monitoring
- If HbA1c rises to 58 mmol/mol or higher on initial medication, reinforce lifestyle and adherence, support a target of 53 mmol/mol, and intensify medicines where appropriate.
- Do not routinely advise self-monitoring of capillary blood glucose unless the person uses insulin, has hypoglycaemic episodes, takes a hypoglycaemia-risk medicine while driving or operating machinery, is pregnant or planning pregnancy, is starting corticosteroids short term, or needs confirmation of suspected hypoglycaemia.
6. SCA Consultation Essentials
- Likely SCA tasks include explaining a new diagnosis, negotiating medicines after rising HbA1c, dealing with metformin intolerance, sick-day safety, or assessing a foot or eye concern.
- Gather the clinical data that changes the plan: symptoms, duration, weight change, intercurrent illness, pregnancy plans, renal function, cardiovascular disease, heart failure, frailty, obesity, hypoglycaemia, driving or machinery work, current medicines, adherence and side effects.
- Pregnancy or planning pregnancy changes the adult pathway: arrange preconception care or immediate joint diabetes/antenatal care, review glucose-lowering and complication medicines, advise contraception until glucose control is optimised, stop oral glucose-lowering drugs before pregnancy except metformin when appropriate, and stop ACE inhibitors/angiotensin-II receptor antagonists and statins before conception or as soon as pregnancy is confirmed.
- Use non-judgemental language around weight and treatment adherence. Explain that type 2 diabetes means insulin is not working well enough, or the body is not making enough insulin, so glucose stays too high in the blood.
- The consultation pivot is: “This is long-term, but the plan is practical — reduce glucose safely, reduce heart and kidney risk, and check early for eye, kidney, nerve and foot complications.”
- Safety-net specifically for vomiting, dehydration, confusion, drowsiness, breathlessness, persistent high glucose, ketones, sudden visual loss, or any new ulcer, infection, colour change, swelling or pain in the feet.
7. Red Flags / Escalation / Referral
- Arrange emergency hospital admission if diabetic ketoacidosis or hyperosmolar hyperglycaemic state is suspected.
- Suspect diabetic ketoacidosis in known diabetes or significant hyperglycaemia with polydipsia, polyuria, weight loss, abdominal pain, nausea or vomiting, shortness of breath, lethargy, drowsiness, confusion, acetone breath, acidotic breathing, tachycardia, dehydration, shock or raised ketones. Test ketones even if glucose is near normal, especially with SGLT-2 inhibitor use.
- Suspect hyperosmolar hyperglycaemic state in an unwell adult with severe hyperglycaemia, typically above 30 mmol/L, over several days, with dehydration, disorientation, confusion or drowsiness, and no significant ketosis.
- During intercurrent illness, arrange immediate admission if there is immediate risk of diabetic ketoacidosis, immediate risk of hyperosmolar hyperglycaemic state, suspected acute kidney injury not manageable in primary care, or an insulin-treated person not improving with insulin treatment.
- Eye escalation: emergency ophthalmology assessment for sudden loss of vision, rubeosis iridis, pre-retinal or vitreous haemorrhage, or suspected retinal detachment. Urgent ophthalmology referral is needed for a large, sudden, unexplained reduction in visual acuity.
- Foot escalation: limb or life-threatening diabetic foot problems need immediate referral to acute services and notification of the multidisciplinary foot care service. Other active diabetic foot problems need referral within 1 working day for triage within a further working day.
- Foot risk after assessment: Low risk — continue annual foot checks.
- Moderate risk, such as deformity, neuropathy or peripheral arterial disease — refer to the foot protection service to be seen within 6–8 weeks.
- High risk, such as previous ulceration or amputation, renal replacement therapy, neuropathy plus peripheral arterial disease, or neuropathy/peripheral arterial disease with callus or deformity — refer to the foot protection service to be seen within 2–4 weeks.
- Active foot problems follow the urgent pathways above.
8. What the GP Should Do Today
- Assess symptoms, duration, hydration, ketone risk, infection, weight change, cardiovascular disease, kidney disease, heart failure, frailty, pregnancy plans, mood and treatment burden.
- Examine blood pressure, weight, body mass index, cardiovascular status where relevant, injection sites if on insulin or injectable therapy, and both feet if any foot symptoms or annual review is due.
- Investigate with HbA1c or plasma glucose as appropriate, and arrange repeat testing if asymptomatic. At diagnosis and review, arrange kidney, lipid, blood pressure, weight, smoking and foot-risk checks. Refer to the local eye screening service at diagnosis and check eye-screening status and attendance at annual review.
- Treat with an individualised plan: structured education, diet and physical activity advice, weight support where appropriate, smoking cessation, alcohol advice, oral health advice, immunisation against influenza and pneumococcal infection where indicated, and medicine choice based on comorbidity.
- Advise carrying diabetes identification. Discuss contraception and pre-pregnancy counselling where relevant.
- Review HbA1c every 3–6 months until stable, then 6-monthly. Review medicines for tolerability, adherence, side effects and whether ineffective medicines should be stopped.
9. Practical Use in GP: How to Apply This Topic
Before confirming diagnosis
- Check symptoms and whether HbA1c is valid.
- Repeat testing if the adult is asymptomatic.
- Do not ignore acute stress hyperglycaemia.
Starting / changing treatment
- Agree an HbA1c target before escalating.
- Start multiple medicines sequentially.
- Check renal function before metformin and SGLT-2 inhibitors.
- Consider cardiovascular, renal, obesity and frailty status before choosing medicines.
Sick-day rules
- Give written, individualised advice.
- Intercurrent illness may worsen hyperglycaemia even if intake is reduced.
- During acute illness with dehydration risk, temporarily stop ACE inhibitors, angiotensin II receptor antagonists, diuretics and NSAIDs to reduce acute kidney injury risk. Restart once the person is feeling better and eating and drinking for 24–48 hours, unless renal function is a concern.
- Temporarily stop metformin if dehydration risk is present.
- With SGLT-2 inhibitors, check ketones and stop treatment if acutely unwell or at risk of dehydration.
- Do not stop insulin; insulin dose may need adjustment with specialist diabetes advice if uncertain.
- Medicines may usually be restarted once the person is better and eating and drinking for 24–48 hours, unless renal function is a concern.
Procedural medicine safety
- For intravascular iodinated contrast, stop metformin before or at the time of imaging and restart no earlier than 48 hours afterwards, once renal function has been rechecked and is stable. Stop metformin at surgery under general, spinal or epidural anaesthesia; restart no earlier than 48 hours after surgery or resumption of oral nutrition, once renal function is stable.
- Interrupt SGLT-2 inhibitors for hospitalised major surgery or acute serious illness, monitor ketones preferably in blood, and restart only when ketones are normal and the person is clinically stable. Do not restart an SGLT-2 inhibitor after DKA unless another cause was identified and resolved.
Self-monitoring and devices
- Do not start routine capillary glucose monitoring for everyone.
- If monitoring is indicated, review skills, frequency, equipment, interpretation, action plans, quality-of-life impact and ongoing benefit at least annually.
- Continuous glucose monitoring should be provided by a team with expertise; capillary testing is still needed for accuracy checks and as backup.
Driving
- Driving: Group 1 drivers must tell DVLA if insulin treatment lasts, or will last, more than 3 months. Group 2 drivers must stop driving and notify DVLA when starting insulin. Severe hypoglycaemia while driving means stop driving and notify DVLA. Hypoglycaemia-risk tablets usually do not require Group 1 notification if DVLA criteria are met, but Group 2 drivers must notify. Insulin-treated drivers should check glucose at the start of the first journey and at least every 2 hours while driving; CGM users must carry a finger-prick blood glucose meter as backup.
10. Medicines, Investigations and Intervention Safety
Metformin
Check renal function before starting. Do not start, and stop, if estimated glomerular filtration rate is below 30 mL/min/1.73 m²; review dose below 45 mL/min/1.73 m². Gastrointestinal effects are common during initiation. Lactic acidosis is rare but potentially life-threatening. Vitamin B12 deficiency is common with higher dose, longer duration or risk factors; test if suspected, for example with megaloblastic anaemia or new neuropathy, and consider periodic monitoring in at-risk people.
For intravascular iodinated contrast, stop metformin before or at the time of imaging and restart no earlier than 48 hours afterwards, once renal function has been rechecked and is stable. Stop metformin at surgery under general, spinal or epidural anaesthesia; restart no earlier than 48 hours after surgery or resumption of oral nutrition, once renal function is stable.
SGLT-2 inhibitors
Examples include canagliflozin, dapagliflozin, empagliflozin and ertugliflozin. Main safety issue: diabetic ketoacidosis, including with near-normal glucose. Check risk before starting: previous diabetic ketoacidosis, intercurrent illness, dehydration or volume depletion, and very low carbohydrate or ketogenic diets. Stop and seek urgent advice if diabetic ketoacidosis symptoms occur.
Do not prescribe SGLT-2 inhibitors in active foot disease such as ulceration, osteomyelitis or gangrene. Use caution with previous foot ulcer, peripheral arterial disease or lower-limb amputation, especially with canagliflozin. Advise urgent assessment and stopping treatment if skin ulceration, discolouration, infection or new foot pain/tenderness develops.
Fournier’s gangrene is rare but serious: severe genital or perineal pain, tenderness, redness or swelling with fever or malaise needs immediate assessment. If Fournier’s gangrene is suspected, stop the SGLT-2 inhibitor immediately and arrange urgent treatment.
Interrupt SGLT-2 inhibitors for hospitalised major surgery or acute serious illness, monitor ketones preferably in blood, and restart only when ketones are normal and the person is clinically stable. Do not restart an SGLT-2 inhibitor after DKA unless another cause was identified and resolved.
Glucagon-like peptide-1 receptor agonists and tirzepatide
Adult pathway examples include liraglutide, dulaglutide and semaglutide; tirzepatide is a dual glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 receptor agonist. Do not combine a glucagon-like peptide-1 receptor agonist or tirzepatide with a dipeptidyl peptidase-4 inhibitor.
For GLP-1 receptor agonists or tirzepatide, advise urgent medical attention for severe persistent abdominal pain that may radiate to the back, especially with nausea or vomiting; stop treatment if pancreatitis is suspected and do not restart if confirmed. For women, trans men and non-binary people of childbearing potential, explain that weight loss may improve fertility, effective contraception is needed while taking treatment, and contraception should continue for the product-specific period after stopping. With tirzepatide, reduced oral contraceptive efficacy cannot be excluded, so a non-oral method or added barrier method is advised when starting and for 4 weeks after each dose escalation.
Counsel on gastrointestinal adverse effects, dehydration risk and aspiration risk around general anaesthesia or deep sedation. Semaglutide requires urgent ophthalmology assessment if sudden vision loss occurs.
Sulfonylureas and insulin
These can cause hypoglycaemia. This matters for falls, frailty, driving, machinery and alcohol. If a glucagon-like peptide-1 receptor agonist is added to insulin or a sulfonylurea, consider reducing the latter to reduce hypoglycaemia risk. Insulin with a glucagon-like peptide-1 receptor agonist should only be offered after specialist advice with consultant-led multidisciplinary support.
Pioglitazone
Avoid in heart failure or history of heart failure, uninvestigated macroscopic haematuria, active or previous bladder cancer, and hepatic impairment. Assess bladder cancer, fracture and heart failure risk before starting. Review safety and efficacy after 3–6 months and stop if response is inadequate.
Cardiovascular prevention
Do not routinely offer aspirin or clopidogrel for primary prevention of cardiovascular disease in adults with type 2 diabetes. Use lipid and blood pressure guidance for primary and secondary prevention; established cardiovascular disease changes statin intensity and diabetes medicine choice.
11. How to Explain It to the Patient
“Type 2 diabetes means glucose is building up in your blood because insulin is not working well enough, or your body is not making enough of it.”
“Our aim is not just to lower the glucose number; it is to reduce your risk of heart, kidney, eye, nerve and foot problems.”
“We will agree a target that is safe for you, because too tight a target can cause problems if it leads to hypos.”
“You do not need routine finger-prick testing unless there is a reason, such as insulin, hypos, driving risk, pregnancy planning or short-term steroid treatment.”
“If you are vomiting, dehydrated, very drowsy, confused, short of breath, or cannot keep fluids down, you need urgent medical advice.”
“Any new foot ulcer, infection, colour change, swelling or sudden eye problem should be checked urgently.”
12. When the Plan Changes
If the adult is asymptomatic with one abnormal HbA1c.
- Why this changes the plan: One result is not enough to diagnose diabetes.
- What the GP does now: Repeat testing, preferably using the same test.
If HbA1c is invalid or unreliable.
- Why this changes the plan: The result may misclassify diagnosis or control.
- What the GP does now: Use fasting plasma glucose for diagnosis or alternative trend measures, and seek specialist diabetes or clinical biochemistry advice if uncertain.
If the person has heart failure, atherosclerotic cardiovascular disease, chronic kidney disease, obesity, early-onset diabetes or frailty.
- Why this changes the plan: Medicine choice changes beyond glucose lowering.
- What the GP does now: Choose treatment using the relevant adult pathway and safety checks.
If pregnancy or pregnancy planning is relevant.
- Why this changes the plan: The routine adult medicine pathway no longer applies safely.
- What the GP does now: Arrange preconception care or immediate joint diabetes/antenatal care and review glucose-lowering and complication medicines.
If the person is acutely unwell, dehydrated, vomiting or ketotic.
- Why this changes the plan: Diabetic ketoacidosis, hyperosmolar hyperglycaemic state or acute kidney injury may be developing.
- What the GP does now: Assess urgently, check ketones where appropriate, apply sick-day rules and arrange admission or urgent specialist advice when criteria are met.
If there is an active diabetic foot problem.
- Why this changes the plan: Delay risks limb- or life-threatening complications.
- What the GP does now: Refer immediately to acute services if limb or life-threatening; otherwise refer within 1 working day.
13. Common AKT / SCA Traps
- Diagnosing asymptomatic type 2 diabetes from one abnormal result.
- Missing diabetic ketoacidosis because glucose is normal or near normal in a person taking an SGLT-2 inhibitor.
- Advising routine capillary glucose monitoring for every adult with type 2 diabetes.
- Combining a glucagon-like peptide-1 receptor agonist or tirzepatide with a dipeptidyl peptidase-4 inhibitor.
- Rapidly reducing or stopping insulin when starting a glucagon-like peptide-1 receptor agonist.
- Treating active diabetic foot disease as routine podiatry.
- Forgetting to check eye-screening status and attendance, rather than assuming annual retinal screening for everyone.
- Forgetting annual checks for kidney, foot, blood pressure, lipid, weight and smoking.
14. Common Exam Angles
- Angle: Newly diagnosed adult with HbA1c 51 mmol/mol and no symptoms.
- Hidden challenge: Diagnosis is not confirmed.
- What the candidate must not miss: Repeat testing is needed.
- Angle: Adult with type 2 diabetes on an SGLT-2 inhibitor, vomiting and abdominal pain, glucose not very high.
- Hidden challenge: Euglycaemic diabetic ketoacidosis risk.
- What the candidate must not miss: Check ketones and arrange urgent assessment.
- Angle: Poor control with heart failure or chronic kidney disease.
- Hidden challenge: Medicine choice is comorbidity-led, not just HbA1c-led.
- What the candidate must not miss: SGLT-2 inhibitor role and renal thresholds.
- Angle: New foot ulcer with fever or ischaemia.
- Hidden challenge: Limb or life-threatening diabetic foot problem.
- What the candidate must not miss: Immediate acute referral.
15. 90 Second Audio Summary Script
Type 2 diabetes in adults is an exam topic where the answer changes with symptoms, HbA1c validity, comorbidity and safety. Diagnose persistent hyperglycaemia using HbA1c, fasting plasma glucose, or random glucose with symptoms, but repeat the test if the adult has no symptoms. Do not use HbA1c for diagnosis when the source says it is unreliable, such as pregnancy, very recent symptoms, acute illness, end-stage renal disease or abnormal red cell states.
Management starts with an individualised care plan, structured education, lifestyle support, complication screening and a safe medicine pathway. HbA1c is usually checked every 3 to 6 months until stable, then every 6 months. Routine finger-prick glucose monitoring is not for everyone; it is for specific situations such as insulin, hypos, driving risk, pregnancy planning, corticosteroids or suspected hypoglycaemia.
Medicine choice is shaped by comorbidity. Modified-release metformin and an SGLT-2 inhibitor are central for many adults, but heart failure, atherosclerotic cardiovascular disease, chronic kidney disease, obesity, early-onset diabetes, frailty and pregnancy planning all change the plan. Do not combine a glucagon-like peptide-1 receptor agonist or tirzepatide with a DPP-4 inhibitor.
The dangerous misses are diabetic ketoacidosis, hyperosmolar hyperglycaemic state, sudden visual loss and active diabetic foot disease. Remember that diabetic ketoacidosis can occur in type 2 diabetes and may present with near-normal glucose, especially with SGLT-2 inhibitors. Give sick-day advice, check eye-screening status and attendance, arrange the annual care processes, and make safety-netting specific.
References
· BMJ Group and Pharmaceutical Press (2026) British National Formulary: Dapagliflozin. Available at: https://bnf.nice.org.uk/drugs/dapagliflozin/ (Accessed: 27 April 2026).
· BMJ Group and Pharmaceutical Press (2026) British National Formulary: Diabetic hyperglycaemic emergencies. Available at: https://bnf.nice.org.uk/treatment-summaries/diabetic-hyperglycaemic-emergencies/ (Accessed: 27 April 2026).
· BMJ Group and Pharmaceutical Press (2026) British National Formulary: Empagliflozin. Available at: https://bnf.nice.org.uk/drugs/empagliflozin/ (Accessed: 27 April 2026).
· BMJ Group and Pharmaceutical Press (2026) British National Formulary: Hypoglycaemia. Available at: https://bnf.nice.org.uk/treatment-summaries/hypoglycaemia/ (Accessed: 27 April 2026).
· BMJ Group and Pharmaceutical Press (2026) British National Formulary: Insulin. Available at: https://bnf.nice.org.uk/treatment-summaries/insulin/ (Accessed: 27 April 2026).
· BMJ Group and Pharmaceutical Press (2026) British National Formulary: Metformin hydrochloride. Available at: https://bnf.nice.org.uk/drugs/metformin-hydrochloride/ (Accessed: 27 April 2026).
· BMJ Group and Pharmaceutical Press (2026) British National Formulary: Semaglutide. Available at: https://bnf.nice.org.uk/drugs/semaglutide/ (Accessed: 27 April 2026).
· BMJ Group and Pharmaceutical Press (2026) British National Formulary: Tirzepatide. Available at: https://bnf.nice.org.uk/drugs/tirzepatide/ (Accessed: 27 April 2026).
· BMJ Group and Pharmaceutical Press (2026) British National Formulary: Type 2 diabetes. Available at: https://bnf.nice.org.uk/treatment-summaries/type-2-diabetes/ (Accessed: 27 April 2026).
· Driver and Vehicle Licensing Agency (2025) Diabetes mellitus: assessing fitness to drive. Last updated 7 November 2025. Available at: https://www.gov.uk/guidance/diabetes-mellitus-assessing-fitness-to-drive (Accessed: 27 April 2026).
· Driver and Vehicle Licensing Agency (2025) Diabetes and driving. Available at: https://www.gov.uk/diabetes-driving (Accessed: 27 April 2026).
· Electronic Medicines Compendium (2024) Metformin hydrochloride 500 mg prolonged-release tablets: Summary of Product Characteristics. Strides Pharma UK Ltd. Last updated 12 June 2024. Available at: https://www.medicines.org.uk/emc/ (Accessed: 27 April 2026).
· Electronic Medicines Compendium (2025) Metformin 500 mg film-coated tablets: Summary of Product Characteristics. Brown & Burk UK Ltd. Last updated 9 April 2025. Available at: https://www.medicines.org.uk/emc/ (Accessed: 27 April 2026).
· Medicines and Healthcare products Regulatory Agency (2014) Pioglitazone: risk of bladder cancer. Drug Safety Update. Published 11 December 2014. Available at: https://www.gov.uk/drug-safety-update/pioglitazone-risk-of-bladder-cancer (Accessed: 27 April 2026).
· Medicines and Healthcare products Regulatory Agency (2016) SGLT2 inhibitors: updated advice on the risk of diabetic ketoacidosis. Drug Safety Update. Published 18 April 2016. Available at: https://www.gov.uk/drug-safety-update/sglt2-inhibitors-updated-advice-on-the-risk-of-diabetic-ketoacidosis (Accessed: 27 April 2026).
· Medicines and Healthcare products Regulatory Agency (2019a) SGLT2 inhibitors: reports of Fournier’s gangrene (necrotising fasciitis of the genitalia or perineum). Drug Safety Update. Published 18 February 2019. Available at: https://www.gov.uk/drug-safety-update/sglt2-inhibitors-reports-of-fournier-s-gangrene (Accessed: 27 April 2026).
· Medicines and Healthcare products Regulatory Agency (2019b) GLP-1 receptor agonists: reports of diabetic ketoacidosis when concomitant insulin was rapidly reduced or discontinued. Drug Safety Update. Published 19 June 2019. Available at: https://www.gov.uk/drug-safety-update/glp-1-receptor-agonists-reports-of-diabetic-ketoacidosis-when-concomitant-insulin-was-rapidly-reduced-or-discontinued (Accessed: 27 April 2026).
· Medicines and Healthcare products Regulatory Agency (2020) SGLT2 inhibitors: monitor ketones in blood during treatment interruption for surgical procedures or acute serious medical illness. Drug Safety Update. Published 18 March 2020. Available at: https://www.gov.uk/drug-safety-update/sglt2-inhibitors-monitor-ketones-in-blood-during-treatment-interruption-for-surgical-procedures-or-acute-serious-medical-illness (Accessed: 27 April 2026).
· Medicines and Healthcare products Regulatory Agency (2022) Metformin and reduced vitamin B12 levels: new advice for monitoring patients at risk. Drug Safety Update. Published 20 June 2022. Available at: https://www.gov.uk/drug-safety-update/metformin-and-reduced-vitamin-b12-levels-new-advice-for-monitoring-patients-at-risk (Accessed: 27 April 2026).
· Medicines and Healthcare products Regulatory Agency (2025) GLP-1 and dual GIP/GLP-1 receptor agonists: potential risk of pulmonary aspiration during general anaesthesia or deep sedation. Drug Safety Update. Published 28 January 2025. Available at: https://www.gov.uk/drug-safety-update/glp-1-and-dual-gip-glp-1-receptor-agonists-potential-risk-of-pulmonary-aspiration-during-general-anaesthesia-or-deep-sedation (Accessed: 27 April 2026).
· Medicines and Healthcare products Regulatory Agency (2026a) GLP-1 receptor agonists and dual GLP-1/GIP receptor agonists: strengthened warnings on acute pancreatitis, including necrotising and fatal cases. Drug Safety Update. Published 29 January 2026. Available at: https://www.gov.uk/drug-safety-update/glp-1-receptor-agonists-and-dual-glp-1-gip-receptor-agonists-strengthened-warnings-on-acute-pancreatitis-including-necrotising-and-fatal-cases (Accessed: 27 April 2026).
· Medicines and Healthcare products Regulatory Agency (2026b) Semaglutide (Wegovy, Ozempic and Rybelsus): risk of non-arteritic anterior ischaemic optic neuropathy (NAION). Drug Safety Update. Published 5 February 2026. Available at: https://www.gov.uk/drug-safety-update/semaglutide-wegovy-ozempic-and-rybelsus-risk-of-non-arteritic-anterior-ischemic-optic-neuropathy-naion (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2015) Suspected cancer: recognition and referral. NICE guideline NG12. Available at: https://www.nice.org.uk/guidance/ng12 (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2019) Diabetic foot problems: prevention and management. NICE guideline NG19. Last updated 11 October 2019. Available at: https://www.nice.org.uk/guidance/ng19 (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2020) Diabetes in pregnancy: management from preconception to the postnatal period. NICE guideline NG3. Last updated 16 December 2020. Available at: https://www.nice.org.uk/guidance/ng3 (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2021) Chronic kidney disease: assessment and management. NICE guideline NG203. Last updated 24 November 2021. Available at: https://www.nice.org.uk/guidance/ng203 (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2023) Cardiovascular disease: risk assessment and reduction, including lipid modification. NICE guideline NG238. Available at: https://www.nice.org.uk/guidance/ng238 (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2024) Diabetic retinopathy: management and monitoring. NICE guideline NG242. Available at: https://www.nice.org.uk/guidance/ng242 (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2025) Tirzepatide for treating type 2 diabetes. Technology appraisal guidance TA924. Last updated 1 September 2025. Available at: https://www.nice.org.uk/guidance/ta924 (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2026a) Type 2 diabetes in adults: management. NICE guideline NG28. Last updated 18 February 2026. Available at: https://www.nice.org.uk/guidance/ng28 (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2026b) Type 2 diabetes in adults. NICE quality standard QS209. Last updated 18 February 2026. Available at: https://www.nice.org.uk/guidance/qs209 (Accessed: 27 April 2026).
· National Institute for Health and Care Excellence (2026c) Hypertension in adults: diagnosis and management. NICE guideline NG136. Last updated 26 February 2026. Available at: https://www.nice.org.uk/guidance/ng136 (Accessed: 27 April 2026).
· NHS (2026) Diabetic eye screening. Available at: https://www.nhs.uk/conditions/diabetic-eye-screening/ (Accessed: 27 April 2026).
· NHS (n.d.) Diabetes and pregnancy. Available at: https://www.nhs.uk/pregnancy/related-conditions/existing-health-conditions/diabetes/ (Accessed: 27 April 2026).
· NHS (n.d.) Metformin. Available at: https://www.nhs.uk/medicines/metformin/ (Accessed: 27 April 2026).
· NHS (n.d.) Type 2 diabetes. Available at: https://www.nhs.uk/conditions/type-2-diabetes/ (Accessed: 27 April 2026).
· NHS England (2026) Diabetic eye screening: programme overview. Last updated 17 March 2026. Available at: https://www.gov.uk/guidance/diabetic-eye-screening-programme-overview (Accessed: 27 April 2026).
· Royal College of General Practitioners (2025) Metabolic problems and endocrinology. RCGP Curriculum Topic Guide. Published 1 August 2025. Available at: https://www.rcgp.org.uk/mrcgp-exams/gp-curriculum (Accessed: 27 April 2026).
· Davies, M.J., et al. (2022) ‘Management of hyperglycaemia in type 2 diabetes, 2022. A consensus report by the American Diabetes Association and the European Association for the Study of Diabetes’, Diabetologia, 65, pp. 1925–1966.
· Diabetes UK (2025) Diabetes in the UK 2025: statistics and facts. London: Diabetes UK. Available at: https://www.diabetes.org.uk/ (Accessed: 27 April 2026).
· GBD 2021 Diabetes Collaborators (2023) ‘Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: a systematic analysis for the Global Burden of Disease Study 2021’, The Lancet, 402(10397), pp. 203–234.
· Schaper, N.C., et al. (2020) ‘Practical guidelines on the prevention and management of diabetic foot disease: IWGDF 2019 update’, Diabetes/Metabolism Research and Reviews, 36(S1), e3266.
· World Health Organization (2019) Classification of diabetes mellitus. Geneva: World Health Organization. Available at: https://www.who.int/publications/i/item/classification-of-diabetes-mellitus (Accessed: 27 April 2026).
Important Disclaimer
This MedDigest MRCGP Topic Essential is an independent educational and revision resource, created to support exam preparation only. It is not a clinical guideline, prescribing resource, or a substitute for your own professional judgment.
This content is designed to highlight exam-relevant clinical principles, management pathways, and consultation approaches in a concise format. Any example explanations, consultation wording, scenario angles, or summary scripts are illustrative and should not be used as stand-alone clinical advice.
This resource has not been produced, reviewed, or endorsed by NICE, the Royal College of General Practitioners, or any other official organisation.
Medicine and guidance change over time. For definitive recommendations, always consult the latest official guidance, the BNF, and your local clinical policies and referral pathways.
MedDigest and its authors accept no responsibility for any loss, harm, or adverse outcome arising from reliance on the information contained in this resource.

 Home
Home